
|
|
|
 | |
 |
 |
|
| ||||
|
| |||||||
|
|
Research is the foundation upon which the Meinwald Group is built. As past, ongoing, and future projects are adapted to web format, they will join the ranks of the research represented on these pages.
Chiral Silylation Reagents for the Determination of Absolute Configuration Using NMR SpectroscopyA common method of defining absolute stereochemistry in small molecules involves derivatizing samples with a chiral reagent and comparing the product with authentic samples of derivatized stereoisomers by either chromatography or NMR spectroscopy. A variety of optically pure reagents have been used to determine absolute stereochemistry in this manner, and the introduction of optically pure (R)- and (S)-alpha-(trifluoromethyl)phenylacetic acid (MTPA) and the corresponding acid chlorides for this purpose has made a particularly important contribution to determining the absolute configuration of natural products. Recently we found ourselves faced with determining the absolute configuration of the heat- and acid-sensitive macyolenes scontaining a bis-allylic ester moiety, which we identfied from the larvae of Pieris rapae, the cabbage fly.  To establish the absolute configuration of these compounds, we converted the mayolenes into the corresponding hydroxy methyl ester 2, with the intention of using the MTPA methodology to compare natural 2 with synthetic, optically pure (R)- and (S)-2. Unfortunately, due to facile elimination of the free or esterified hydroxyl group in 2 to form a mixture of conjugated tetraenes, we were unable to use MTPA to determine this sensitive alcohol's absolute stereochemistry. Similar difficulties in preparing useful esters with MTPA have been encountered in the determination of the absolute configuration of N-(17-hydroxylinolenoyl)-L-glutamine (volicitin, 3), an important elicitor of "plant volatile" biosynthesis recently identified from the saliva of beet armyworms. 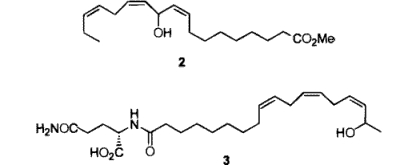 Since MTPA and similar chiral ester-forming reagents are likely to prove unsuitable for the characterization of optically active alcohols which are prone to undergo elimination, we considered the use of chiral silylating reagents for this purpose. Silylations can be carried out under very mild conditions, and the resulting silyl ethers would be expected to be much more resistant to elimination than the corresponding O-acyl derivatives. Furthermore, silyl ethers are easily cleaved, allowing recovery of the original alcohol. As our chiral silylating agent of choice, we prepared (+)- and (-)-chloromenthoxydiphenylsilanes from commercially available dichlorodiphenylsilane and optically pure (+)- and (-)-menthol. Reaction of (+)- and (-)-chloromenthoxydiphenylsilanes with synthetic (11R)-hydroxyoctadeca-9(Z),12(Z),15(Z)-trienoic acid methyl ester 2 with (-)-silane produced the diastereomeric siloxanes 5 and 6 in good yield. 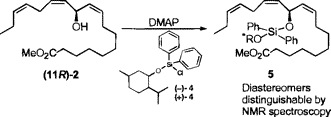
Derivatization of (11R)-2 with a chiral silane. As anticipated, the 1H NMR spectra of these products showed significant differences in their chemical shift patterns, which could be applied toward determining the absolute configuration of the mayolenes. 
1H NMR spectra of siloxane diastereomers 5 and 6 (500 MHz, CDCl3). In addition to the example presented here, several chiral aliphatic alcohols were derivatized, including (17R)-hydroxyoctadeca-9(Z),12(Z),15(Z)-trienoic acid methyl ester, related to volicitin 3. Again, the 1H NMR spectra of the resulting diastereomeric siloxanes showed significant differences, indicating the general applicability of our method. It should be noted that even in cases where proton NMR signals partially overlap, commonly available software packages for the analysis of NMR data can be used to deconvolute the signals of interest, making determination of ee's and absolute configuration feasible. In conclusion, we have prepared chiral silylation reagents that can be used for the determination of absolute configuration and enantiomeric excess of labile chiral alcohols. In the examples we have investigated, stable diastereomeric siloxane derivatives are obtained; their 1H NMR spectra clearly allow us to distinguish between the enantiomers of alcohols from which they are derived. Derivatization conditions are mild and have been optimized for microscale reactions. While our current investigations have mostly focused on the utility of this procedure for the determination of the absolute configuration of secondary, we anticipate that this method can be extended easily to a variety of other structures. For a more detailed description of our investigations and experimental details, please see our recent publication in Organic Letters: Chiral Silylation Reagents for the Determination of Absolute Configuration by NMR Spectroscopy. Douglas B. Weibel, Tameka R. Walker, Frank C. Schroeder, Jerrold Meinwald, Organic Letters 2000, 2, 2381-2383. |